Did you know?
Absolute zero (−273.15°C) is the coldest possible temperature — at which atoms nearly stop moving.
Did you know?
Absolute zero (−273.15°C) is the coldest possible temperature — at which atoms nearly stop moving.
Consider the following compounds below. Hyperconjugation occurs in
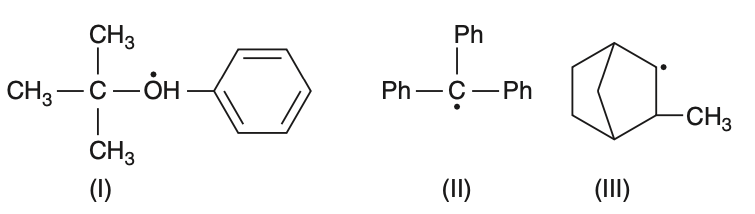
II only
III only
I and III
I only
To determine where hyperconjugation occurs, we need to analyze each compound.Hyperconjugation involves the delocalization of electrons from a -bond (usually C-H) to an adjacent empty or partially filled p-orbital or -orbital.Let's examine each compound:• Compound (I):This is a tertiary carbocation with a benzene ring. The presence of C-H bonds adjacent to the carbocation allows hyperconjugation.• Compound (II):This is a triphenylmethyl radical. The radical is stabilized by resonance with the phenyl groups, not by hyperconjugation.• Compound (III):This is a cyclohexyl radical with a methyl group. The C-H bonds adjacent to the radical can participate in hyperconjugation.Therefore, hyperconjugation occurs in compounds (I) and (III).The correct answer is Option 3: I and III. However, the provided correct option is 2, indicating a possible error in the question or options.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day