Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
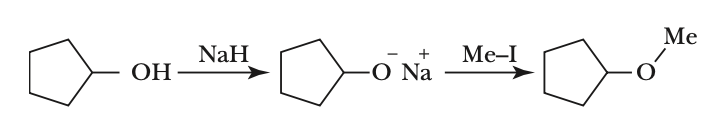
To classify the given reaction, let's analyze the steps involved:1. The first step involves the reaction of cyclopentanol with sodium hydride (NaH):• Cyclopentanol (a primary alcohol) reacts with NaH to form sodium cyclopentoxide and hydrogen gas.• This is an acid-base reaction where the hydroxyl group (OH) is deprotonated.2. The second step involves the reaction of sodium cyclopentoxide with methyl iodide (Me-I):• Sodium cyclopentoxide acts as a nucleophile and attacks the methyl iodide.• This results in the formation of cyclopentyl methyl ether and sodium iodide.The overall reaction is:CyclopentanolSodium cyclopentoxideCyclopentyl methyl etherThis sequence of reactions is known as the Williamson ether synthesis.• The Williamson ether synthesis is a method for preparing ethers by reacting an alkoxide ion with a primary alkyl halide.Therefore, the correct classification of the reaction is:Option 1: Williamson ether synthesis reaction.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day