Did you know?
A bolt of lightning heats the surrounding air to ~30,000 K — five times hotter than the Sun's surface.
Did you know?
A bolt of lightning heats the surrounding air to ~30,000 K — five times hotter than the Sun's surface.
An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric, and isochoric. The curve which represents the adiabatic process among 1, 2, 3 and 4 is:
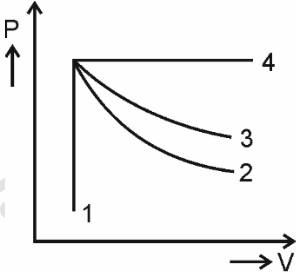
To solve this problem, we need to identify the curve that represents the adiabatic process.In a PV diagram:• An adiabatic process is steeper than an isothermal process.• An isothermal process follows constant.• An adiabatic process follows constant, where Analyzing the given curves:• Curve 1 is vertical, indicating an isochoric process (constant volume).• Curve 4 is horizontal, indicating an isobaric process (constant pressure).• Curves 2 and 3 are downward sloping, indicating either isothermal or adiabatic processes.Since the adiabatic process is steeper than the isothermal process,curve 2 is steeper than curve 3.Therefore, curve 2 represents the adiabatic process.The correct option is 2.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day