Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
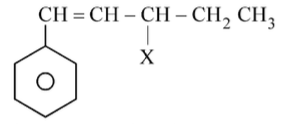
The given compound CH=CH−CH−CH_2−CH_3 | X Is an example of____
Benzylic halide
Aryl halide
Allylic halide
Vinylic halide
To solve this problem, we need to identify the type of halide in the given compound.The structure provided is:• A benzene ring with a side chain: CH=CH-CH-CH-CH with a halogen (X) attached to the third carbon.Let's analyze the options:1. Benzylic halide: A halogen attached to a carbon adjacent to a benzene ring.2. Aryl halide: A halogen directly attached to a benzene ring.3. Allylic halide: A halogen attached to a carbon adjacent to a carbon-carbon double bond.4. Vinylic halide: A halogen attached to a carbon that is part of a carbon-carbon double bond.In the given compound, the halogen (X) is attached to a carbon that is adjacent to a carbon-carbon double bond.This fits the definition of an allylic halide.Therefore, the correct option is Option 3: Allylic halide.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day