Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
Did you know?
The observable universe is ~93 billion light-years across — and it's still expanding.
Given the three structures given below:The enthalpy of hydrogenation of these compounds will be in the order as
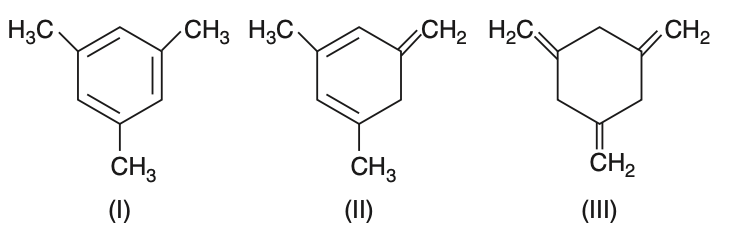
To determine the order of enthalpy of hydrogenation, we need to consider the stability of the compounds.Given structures:• Structure (I) is 1,2,3-trimethylbenzene (aromatic compound).• Structure (II) is 1,2,3-trimethylcyclohexene (non-aromatic with one double bond).• Structure (III) is 1,3,5-hexatriene (non-aromatic with three double bonds).The enthalpy of hydrogenation is inversely related to the stability of the compound.Aromatic compounds are more stable due to resonance, resulting in lower enthalpy of hydrogenation.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day