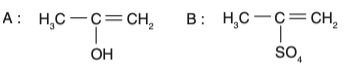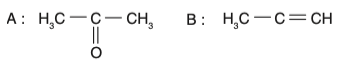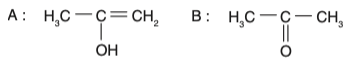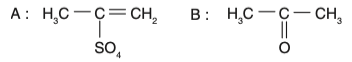0days until NEET 2026Every question counts. ✊
🫀
Did you know?
Your heart beats ~100,000 times a day — roughly 3 billion beats in a lifetime.
Predict the correct intermediate and product in the following reaction: H3C−C≡CHH2O,H2SO4HgSO4 Intermediate A→ Product B
hard
Hydrocarbons
2017
chemistry
Explanation
To solve this problem, we need to understand the reaction mechanism of the hydration of alkynes.The reaction given is: H3C−C≡CHH2O,H2SO4HgSO4
Our AI powered practice platform can help you achieve your doctor dream.
Practice 2000+ previous year NEET questionsMore practice, more score
AI generated hintsUse hints to get start solving
AI companion chat to clear doubts 24*7Ask any question, get instant answers
AI generated solutionsGet detailed step by step solutions
Check related NCERT contentRead while solving
Track your progressImprove every day
Sign up / Login Intermediate
Product
This is an example of the hydration of an alkyne, specifically acetylene, in the presence of
mercury(II) sulfate and sulfuric acid, which leads to the formation of a ketone.
Step-by-step mechanism:
1. Formation of the enol (Intermediate A):
• The alkyne reacts with water in the presence of
and
H2SO4. • This leads to the formation of an enol, which is an alcohol with a double bond.
• The enol formed is unstable and quickly tautomerizes to a ketone.
2. Tautomerization to form the ketone (Product B):
• The enol undergoes keto-enol tautomerism, where the hydroxyl group (OH) and the double bond
rearrange to form a carbonyl group (C=O).
• This results in the formation of acetone, a ketone.
Analyzing the options:
• Option 1: The intermediate is an enol, but the product is incorrect.
• Option 2: The intermediate is a ketone, which is incorrect.
• Option 3: The intermediate is an enol, and the product is a ketone (acetone). This is correct.
• Option 4: The intermediate is incorrect.
Therefore, the correct option is Option 3.
Intermediate A:
H3C−C(OH)=CH2 Product B:
H3C−C(=O)−CH3