Did you know?
You have more bacterial cells than human cells — ~38 trillion bacteria vs ~30 trillion human cells.
Did you know?
You have more bacterial cells than human cells — ~38 trillion bacteria vs ~30 trillion human cells.
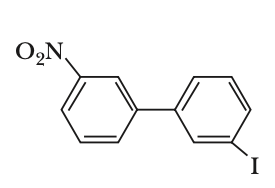
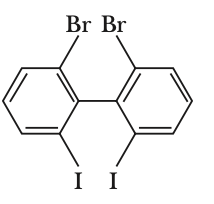
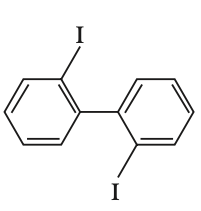
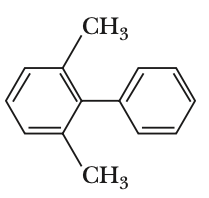
To determine which biphenyl compound is optically active, we need to consider the symmetry and steric hindrance of each compound.Optical activity arises when a compound lacks a plane of symmetry and has a chiral center or axis. In biphenyls, this can occur when the two phenyl rings are twisted out of plane due to steric hindrance, preventing free rotation.Let's analyze each option:Option 1:The compound has a nitro group and an iodine atom. The presence of the nitro group can cause steric hindrance, but the compound is not necessarily chiral.Option 2:The compound has two bromine atoms ortho to the biphenyl bond. This creates significant steric hindrance, preventing the two phenyl rings from being coplanar. This lack of coplanarity can lead to chirality due to restricted rotation, making the compound optically active.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day