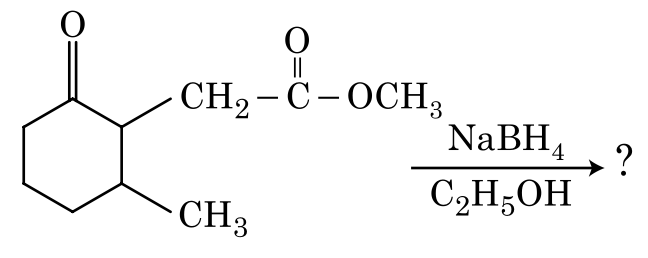0 days until NEET 2026 Every question counts. ✊
🩸
Did you know?
Red blood cells have no nucleus, making more room to carry oxygen-binding haemoglobin.
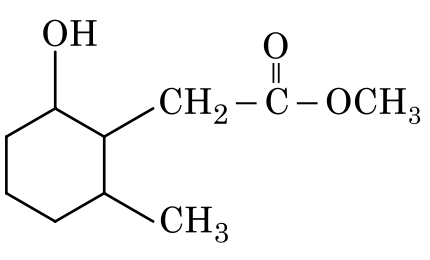
The product formed in the following chemical reaction is: hard
Aldehydes, Ketones and Carboxylic Acids
2021
chemistry
Explanation To solve this problem, we need to understand the reaction taking place. \\
The given reaction involves the reduction of a compound using N a B H 4 \mathrm{NaBH_4} NaB H 4 in C 2 H 5 O H . \mathrm{C_2H_5OH}. \\
C 2 H 5 OH .
Our AI powered practice platform can help you achieve your doctor dream. Practice 2000+ previous year NEET questions More practice, more score
AI generated hints Use hints to get start solving
AI companion chat to clear doubts 24*7 Ask any question, get instant answers
AI generated solutions Get detailed step by step solutions
Check related NCERT content Read while solving
Track your progress Improve every day
Sign up / Login
Let's analyze the structure of the reactant:
• The reactant has two carbonyl groups: a ketone and an ester.
Sodium borohydride (NaBH
) is a selective reducing agent that typically reduces aldehydes and ketones,
but not esters under normal conditions.
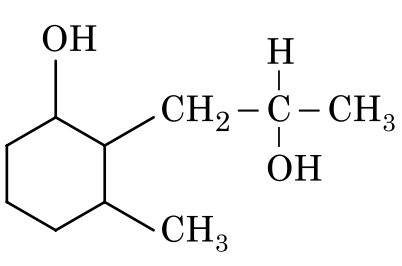
Therefore, in this reaction, only the ketone group will be reduced to an alcohol.
Let's identify the product:
• The ketone group in the cyclohexane ring will be reduced to a secondary alcohol.
• The ester group will remain unchanged.
Now, let's compare with the options:
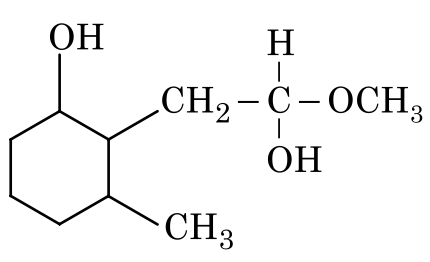
Option 1: Both the ketone and ester are reduced, which is incorrect.
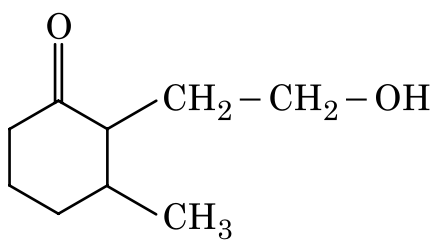
Option 2: The ester is reduced, which is incorrect.
Option 3: Both the ketone and ester are reduced, which is incorrect.
Option 4: Only the ketone is reduced, which is correct.
Therefore, the correct product is shown in Option 4.