0 days until NEET 2026 Every question counts. ✊
🌌
Did you know?
If you removed all empty space from atoms in your body, you'd compress to the size of a speck of dust.
The compound that will undergo Sₙ1 reaction with the fastest rate is medium
Haloalkanes and Haloarenes
2024
chemistry
Explanation To solve this problem, we need to determine which compound undergoes an S N 1 S_N1 S N 1 reaction at the fastest rate.
\newline The S N 1 S_N1 S N 1 reaction rate depends on the stability of the carbocation formed after the leaving group departs.
\newline
Our AI powered practice platform can help you achieve your doctor dream. Practice 2000+ previous year NEET questions More practice, more score
AI generated hints Use hints to get start solving
AI companion chat to clear doubts 24*7 Ask any question, get instant answers
AI generated solutions Get detailed step by step solutions
Check related NCERT content Read while solving
Track your progress Improve every day
Sign up / Login
Let's analyze each option:
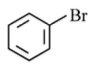
Option 1:
• The compound is bromobenzene.
• The leaving group is directly attached to the benzene ring.
• Aryl halides do not form stable carbocations, so
is unlikely.
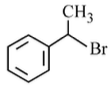
Option 2:
• The compound is 1-phenylethyl bromide.
• The leaving group is attached to a secondary carbon adjacent to a phenyl group.
• The resulting carbocation is benzylic, which is highly stabilized by resonance with the phenyl ring.
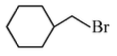
Option 3:
• The compound is cyclohexylmethyl bromide.
• The leaving group is attached to a primary carbon.
• Primary carbocations are not stable, so
is less favorable.
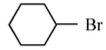
Option 4:
• The compound is cyclohexyl bromide.
• The leaving group is attached to a secondary carbon.
• The resulting carbocation is secondary, which is more stable than primary but less stable than benzylic.
Conclusion:
• Option 2 forms a benzylic carbocation, which is the most stable among the options.
• Therefore, the compound in Option 2 will undergo the
reaction at the fastest rate.
This corresponds to Option 2.