Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
Did you know?
Diamonds and graphite are both made of pure carbon — just arranged differently.
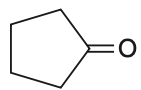
Treatment of cyclopentanone O with methyl lithium gives which of the following species?
Cyclopentanonyl cation
Cyclopentanonyl radical
Cyclopentanonyl biradical
Cyclopentanonyl anion
To solve this problem, we need to understand the reaction between cyclopentanone and methyl lithium.Cyclopentanone is a ketone, and methyl lithium (CHLi) is an organolithium reagent that acts as a strong nucleophile.The reaction proceeds as follows:1. The carbonyl group () in cyclopentanone is polarized, with the carbon being electrophilic.2. Methyl lithium provides a methyl anion (CH) which attacks the electrophilic carbon of the carbonyl group.3. This nucleophilic attack opens the carbonyl group, forming an alkoxide intermediate.The reaction can be represented as:Cyclopentanone + CHLiCyclopentanonyl anion (alkoxide)The resulting species is an alkoxide ion, which is a type of anion.Therefore, the correct answer is: Cyclopentanonyl anion.This corresponds to Option 4.
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day