0 days until NEET 2026 Every question counts. ✊
🌿
Did you know?
A single strand of DNA, if uncoiled, would be ~6 feet long — yet it fits inside a cell nucleus.
Which one is a wrong statement?
hard
Structure of Atom
2018
chemistry
The electronic configuration of N atom is
An orbital is designated by three quantum numbers while an electron in an atom is designated by four quantum numbers.
Total orbital angular momentum of electron in s s s orbital is equal to zero.
The value of m m m for d z 2 d_{z^2} d z 2 is zero.
Explanation To determine the wrong statement, let's analyze each option: \\
Option 1: The electronic configuration of N atom is \\
The electronic configuration of nitrogen (N) is 1 s 2 2 s 2 2 p 3 . 1s^2 \, 2s^2 \, 2p^3. \\
1 s 2 2 s 2 2 p 3 .
Our AI powered practice platform can help you achieve your doctor dream. Practice 2000+ previous year NEET questions More practice, more score
AI generated hints Use hints to get start solving
AI companion chat to clear doubts 24*7 Ask any question, get instant answers
AI generated solutions Get detailed step by step solutions
Check related NCERT content Read while solving
Track your progress Improve every day
Sign up / Login
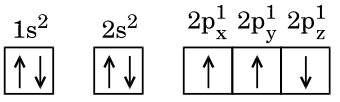
The image shows:
1 s 2 : ↑ ↓ 2 s 2 : ↑ ↓ 2 p x 1 : ↑ 2 p y 1 : ↑ 2 p z 1 : ↑ \\
1s^2: \uparrow\downarrow \quad 2s^2: \uparrow\downarrow \quad 2p_x^1: \uparrow \quad 2p_y^1: \uparrow \quad 2p_z^1: \uparrow \\
1 s 2 :↑↓ 2 s 2 :↑↓ 2 p x 1 :↑ 2 p y 1 :↑ 2 p z 1 :↑ This configuration is correct according to Hund's rule, which states that electrons fill degenerate orbitals singly before pairing.
Thus, Option 1 is correct.
Option 2: An orbital is designated by three quantum numbers while an electron in an atom is designated by four quantum numbers.
• An orbital is described by three quantum numbers:
and
• An electron is described by four quantum numbers:
and
This statement is correct.
Option 3: Total orbital angular momentum of electron in
orbital is equal to zero.
• For an
orbital,
• Orbital angular momentum
L = l ( l + 1 ) ℏ = 0 ( 0 + 1 ) ℏ = 0. L = \sqrt{l(l+1)}\hbar = \sqrt{0(0+1)}\hbar = 0. \\
L = l ( l + 1 ) ℏ = 0 ( 0 + 1 ) ℏ = 0. This statement is correct.
Option 4: The value of
for
is zero.
• The
orbital is one of the five
orbitals, and it has
This statement is correct.
Conclusion:
All statements are correct, but since the question asks for the wrong statement,
we must identify the mistake in the question itself.
The provided electronic configuration in Option 1 is correct, so the question might have an error.
Therefore, the intended answer is Option 1, but it is not actually wrong.