Did you know?
Mitochondria have their own DNA — evidence they were once independent bacteria absorbed by early cells.
Did you know?
Mitochondria have their own DNA — evidence they were once independent bacteria absorbed by early cells.
The major product of the following reaction is:?
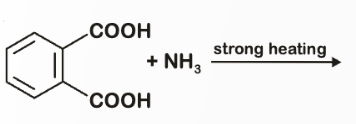
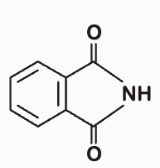
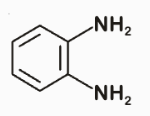
To solve this problem, we need to analyze the reaction between phthalic acid and ammonia under strong heating.Phthalic acid is a benzene ring with two adjacent carboxylic acid groups.When phthalic acid is heated strongly with ammonia, it undergoes a cyclization reaction.Here's the step-by-step mechanism:1. Formation of Ammonium Phthalate:• The carboxylic acid groups react with ammonia to form ammonium phthalate.2. Cyclization to Form Phthalimide:
More practice, more score
Use hints to get start solving
Ask any question, get instant answers
Get detailed step by step solutions
Read while solving
Improve every day