0 days until NEET 2026 Every question counts. ✊
⚡
Did you know?
Light from the Sun takes ~8 minutes 20 seconds to reach Earth, travelling at 300,000 km/s.
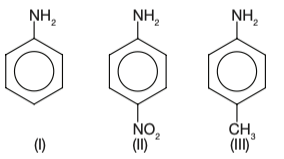
The correct increasing order of basic strength for the following compound is:
medium
Amines
2017
chemistry
I I I < I < I I III < I < II III < I < II
I I I < I I < I III < II < I III < II < I
I I < I < I I I II < I < III II < I < III
I I < I I I < I II < III < I II < III < I
Explanation To determine the increasing order of basic strength for the given compounds, we need to analyze the effect of substituents on the basicity of the amino group ( N H 2 ) . (NH_2).
\newline ( N H 2 ) . The compounds are:
\newline (I) Aniline ( C 6 H 5 N H 2 ) (C_6H_5NH_2)
\newline ( C 6 H 5 N H 2 )
Our AI powered practice platform can help you achieve your doctor dream. Practice 2000+ previous year NEET questions More practice, more score
AI generated hints Use hints to get start solving
AI companion chat to clear doubts 24*7 Ask any question, get instant answers
AI generated solutions Get detailed step by step solutions
Check related NCERT content Read while solving
Track your progress Improve every day
Sign up / Login
(II) p-Nitroaniline
( C 6 H 4 ( N O 2 ) N H 2 ) (C_6H_4(NO_2)NH_2)
\newline ( C 6 H 4 ( N O 2 ) N H 2 ) (III) p-Toluidine
( C 6 H 4 ( C H 3 ) N H 2 ) (C_6H_4(CH_3)NH_2)
\newline ( C 6 H 4 ( C H 3 ) N H 2 ) Basicity is influenced by the electron-donating or electron-withdrawing nature of substituents:
• Electron-donating groups increase basicity by increasing electron density on the nitrogen atom.
• Electron-withdrawing groups decrease basicity by decreasing electron density on the nitrogen atom.
Analysis of substituents:
1. In compound (I), there are no additional substituents, so it serves as a reference.
2. In compound (II), the nitro group
is a strong electron-withdrawing group, reducing the basicity of the amino group.
3. In compound (III), the methyl group
is an electron-donating group, increasing the basicity of the amino group.
Order of basic strength:
• Compound (II) has the lowest basicity due to the electron-withdrawing nitro group.
• Compound (I) has moderate basicity as there are no additional substituents.
• Compound (III) has the highest basicity due to the electron-donating methyl group.
Therefore, the correct increasing order of basic strength is:
Option 3:
I I < I < I I I II < I < III II < I < III